Hapticity in Denial — 3rd April 2017
The following blog is reproduced from http://crimmingroup.org/blog/blog.html
Visit the original for the proper chemical formula formatting etc.
Comparison of the solid-state structures of [UCp4] and [ZrCp4] provides a textbook example of the effect of metal ion size on coordination geometries.[1,2] While both metals are in the +4 oxidation state, U is significantly larger than Zr with covalent radii of 1.96(7) and 1.75(7) Å respectively.[3] As a result, although U4+ is large enough for all the Cp rings to bind eta-5, zirconium is not, and one ring slips to the eta-1 coordination mode. During our work to develop new methods to break carbon–oxygen bonds in ethers we applied an indenyl (Ind = C9H7) analogue of a zirconocene complex as a catalyst. [Zr(η5-Ind)2Cl2] was used in combination with an aluminium reagent for the C–O bond functionalization of benzofuran.[4] Here we present the crystal structure of [Zr(η5-Ind)2(η1-Ind)2Cl] (1), which was serendipitously isolated during preparations of [Zr(η5-Ind)2Cl2] (Figure 1).
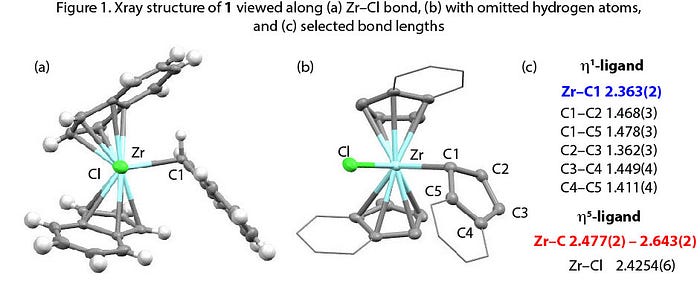
Synthesis of 1 has previously been reported by Alt and co-workers,[5] but crystallographic analysis was not reported. “Sterically-overloaded” 1 and its Hf analogue (1-Hf) were characterised by 1H NMR spectroscopy, showing η1-η5 fluxionality of the indenyl ligand in solution. 1-Hf has been characterised by single crystal diffraction experiment but the quality of the data only allowed conclusion on the general topology. This connectivity is analogous to our structure for 1. Quality crystal growth has also been difficult with the smaller Cp analogue.[6,7] However, X-ray data has been reported once, in the PhD thesis of Strittmatter,[8] whose results show a [Zr(η5-Cp)3Cl] structure. Thus, we infer from comparison with 1 that the Zr centre is sensitive to the steric demands of the L2X-type ligand in these complexes.
Selected bond lengths for 1 are presented above and as primary data through the CCDC (1541881). The hydrogen-atom attached to C1 was found in the Fourier difference density map. Just as with [MCp4] (M = U, Zr) it appears the Zr centre is too crowded to accommodate all three η5-ligands, and one indenyl group slips to an η1 configuration. The crystal structure of 1-U reiterates this point: the U4+ centre is able to bear three indenyl groups as η5-ligands, in contrast to 1.[9] However, changing the X-type ligand on M4+ from a chloride to a hydride results in a different molecular structure (2), as shown by Takahashi and colleagues.[10] An effect of this kind for Cp3MX complexes (M = Zr, Hf) was predicted computationally by Bursten & Palmer in 2006,[11] based upon π-donor ability of the X-type ligand. However, the study considered only η1 or η5 coordination isomers: Takahashi and co-workers’ report of 2 found all three indenyl ligands as η3-coordinated.[10] The results of the X-ray structures for 1, 1-U and 2 show that both metal centre and ancillary ligands have a telling effect on the hapticity of the indenyl moiety. Crystal packing effects may also be in play.
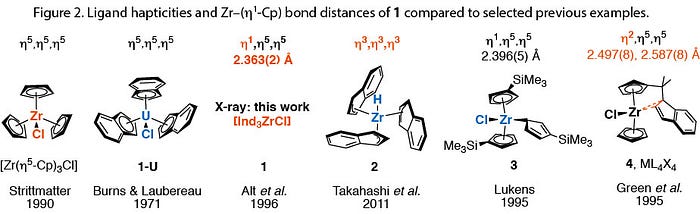
In the crystal structure of 1, a short Zr–C1 distance of 2.363(2) Å is seen. The hitherto shortest (η1-Cp) bond length — 2.396(5) Å in [Zr(η1-CpR)(η5-CpR)2Cl] (3: R = SiMe3).[12,13] In other the Zr–C(η1) containing complexes of these ligands the distances are in excess of 2.447 Å.[13,14]
Finally, we can compare complexes 1–3 to the interesting case of 4 (Figure 2), which contains Ind in an unusual η2 configuration.[15] The latter complex bears a mixed Cp/Ind ligand system, created by Green and co-workers by attaching an indenyl to a cyclopentadienyl moiety via a C1 pendant arm.[16] The bidentate ligand is dianionic, but ligation through the indenyl fragment appears to be a frustrated compromise between η1- and η2 — the Zr–C bond distances being much longer than those in 1–3. In summary, analysis of the structure of 1 in the context of similar structures has borne testament to the known effects that the metal identity, ligand sterics and electronics can have on the hapticity of L2X ligands like Cp and Ind.
- V. I. Kulishov, E. M. Brainina, N. G. Bokiy and Y. T. Struchkov, J. Chem. Soc., Chem. Commun., 1970, 475–475.
- R. D. Rogers, R. V. Bynum and J. L. Atwood, J. Am. Chem. Soc., 1978, 100, 5238–5239.
- B. Cordero, V. Gomez, A. E. Platero-Prats, M. Reves, J. Echeverria, E. Cremades, F. Barragan and S. Alvarez, Dalton Trans., 2008, 2832–2838.
- S. Yow, A. J. P. White, A. E. Nako, L. Neveu and M. R. Crimmin, Organometallics, 2013, 32, 5260–5262.
- C. Schmid, H. G. Alt and W. Milius, J. Organomet. Chem., 1997, 544, 139–142.
- F. Calderazzo, U. Englert, G. Pampaloni and G. Tripepi, J. Organomet. Chem., 1998, 555, 49–56.
- E. J. Palmer, R. J. Strittmatter, K. T. Thornley, J. C. Gallucci and B. E. Bursten, Polyhedron, 2013, 58, 120–128.
- R. J. Strittmatter, PhD Thesis, Ohio State University, 1990.
- J. H. Burns and P. G. Laubereau, Inorg. Chem., 1971, 10, 2789–2792.
- S. Ren, T. Seki, D. Necas, H. Shimizu, K. Nakajima, K.-I. Kanno, Z. Song and T. Takahashi, Chem. Lett., 2011, 40, 1443–1444.
- E. J. Palmer and B. E. Bursten, Polyhedron, 2006, 25, 575–584.
- W. Lukens, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1995, 51, 10–12.
- F. M. Chadwick, R. T. Cooper, A. E. Ashley, J.-C. Buffet and D. M. O’Hare, Organometallics, 2014, 33, 3775–3785, and references therein.
- R. D. Rogers, R. V. Bynum and J. L. Atwood, J. Am. Chem. Soc., 1978, 100, 5238–5239.
- G. M. Diamond, M. L. H. Green, N. A. Popham and A. N. Chernega, J. Chem. Soc., Chem. Commun., 1994, 7, 727–728.
- G. M. Diamond, M. L. H. Green, P. Mountford, N. A. Popham and A. N. Chernega, J. Chem. Soc., Chem. Commun., 1994, 92, 103–105.